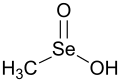
Struktur dan sifat
Asam metanaseleninat telah dikarakterisasi melalui kristalografi sinar-X.[4] Konfigurasi atom selenium berbentuk piramida, dengan Se-C = 1,925(8) Å, Se-O = 1,672(7) Å, Se-OH = 1,756(7) Å, sudut OSeO = 103,0(3)°, sudut HO-Se-C = 93,5(3)°, dan sudut OSeC = 101,4(3)°. Strukturnya isomorf dengan asam metanasulfinat.[5]
Asam benzenaseleninat (C6H5−Se(=O)−OH) sebelumnya telah dikarakterisasi melalui metode sinar-X[6] dan resolusi optiknya telah dilaporkan.[7]
Referensi
- ↑ Ten Brink, Gerd-Jan; Fernandes, Bruno C. M.; Van Vliet, Michiel C. A.; Arends, Isabel W. C. E.; Sheldon, Roger A. (2001). "Selenium catalysed oxidations with aqueous hydrogen peroxide. Part I: Epoxidation reactions in homogeneous solution". Journal of the Chemical Society, Perkin Transactions 1 (3): 224–228. doi:10.1039/b008198l.
- ↑ 1</sup>"},"date":{"wt":"2001"},"last1":{"wt":"Ten Brink"},"first1":{"wt":"Gerd-Jan"},"last2":{"wt":"Vis"},"first2":{"wt":"Jan-Martijn"},"last3":{"wt":"Arends"},"first3":{"wt":"Isabel W. C. E."},"last4":{"wt":"Sheldon"},"first4":{"wt":"Roger A."},"journal":{"wt":"The Journal of Organic Chemistry"},"volume":{"wt":"66"},"issue":{"wt":"7"},"pages":{"wt":"2429–2433"},"pmid":{"wt":"11281784"}},"i":0}}]}' id="mwWQ"/>Ten Brink, Gerd-Jan; Vis, Jan-Martijn; Arends, Isabel W. C. E.; Sheldon, Roger A. (2001). "Selenium-Catalyzed Oxidations with Aqueous Hydrogen Peroxide. 2. Baeyer−Villiger Reactions in Homogeneous Solution1". The Journal of Organic Chemistry. 66 (7): 2429–2433. doi:10.1021/jo0057710. PMID 11281784.
- ↑ Mercier, Eric A.; Smith, Chris D.; Parvez, Masood; Back, Thomas G. (2012). "Cyclic Seleninate Esters as Catalysts for the Oxidation of Sulfides to Sulfoxides, Epoxidation of Alkenes, and Conversion of Enamines to α-Hydroxyketones". The Journal of Organic Chemistry. 77 (7): 3508–3517. doi:10.1021/jo300313v. PMID 22432805.
- ↑ Block, Eric; Birringer, Marc; Jiang, Weiqin; Nakahodo, Tsukasa; Thompson, Henry J.; Toscano, Paul J.; Uzar, Horst; Zhang, Xing; Zhu, Zongjian (2001). "Allium Chemistry: Synthesis, Natural Occurrence, Biological Activity, and Chemistry of Se -Alk(en)ylselenocysteines and Their γ-Glutamyl Derivatives and Oxidation Products". Journal of Agricultural and Food Chemistry. 49 (1): 458–470. doi:10.1021/jf001097b. PMID 11305255.
- ↑ Seff, K.; Heidner, E. G.; Meyers, M.; Trueblood, K. N. "The crystal and molecular structure of methanesulfinic acid." Acta Crystallographica Section B 1969, 25, 350–354.
- ↑ Bryden, J. H.; McCullough, J. D. (1954). "The crystal structure of benzeneseleninic acid". Acta Crystallographica. 7 (12): 833–838. Bibcode:1954AcCry...7..833B. doi:10.1107/S0365110X54002551.
- ↑ 3.0.CO;2-Q"},"title":{"wt":"Optically Active Seleninic Acids: Optical Resolution and Stability"},"date":{"wt":"2001"},"last1":{"wt":"Shimizu"},"first1":{"wt":"Toshio"},"last2":{"wt":"Watanabe"},"first2":{"wt":"Itaru"},"last3":{"wt":"Kamigata"},"first3":{"wt":"Nobumasa"},"journal":{"wt":"Angewandte Chemie International Edition"},"volume":{"wt":"40"},"issue":{"wt":"13"},"pages":{"wt":"2460–2462"},"pmid":{"wt":"11443664"}},"i":0}}]}' id="mwkw"/>Shimizu, Toshio; Watanabe, Itaru; Kamigata, Nobumasa (2001). "Optically Active Seleninic Acids: Optical Resolution and Stability". Angewandte Chemie International Edition. 40 (13): 2460–2462. doi:10.1002/1521-3773(20010702)40:13<2460::AID-ANIE2460>3.0.CO;2-Q. PMID 11443664.
|
|---|
Hanya karbon,
hidrogen
dan oksigen | |
|---|
Hanya satu
unsur
selain
C, H, O | | Nitrogen | |
|---|
| Fosfor | |
|---|
| Sulfur | |
|---|
| Selenium | |
|---|
| Telurium | |
|---|
|
|---|
| Lainnya | |
|---|
|
|