Drospirenon adalah obat progestin dan antiandrogen yang digunakan dalam pil kontrasepsi oral gabungan untuk mencegah kehamilan dan terapi hormon menopause. Obat ini tersedia baik sebagai sediaan tunggal maupun dalam kombinasi dengan etinilestradiol. Obat ini merupakan analog dari spironolakton. Obat ini diminum.
Sumber: Lihat artikel asli di Wikipedia

 | |
 | |
| Data klinis | |
|---|---|
| Pengucapan | Droe-SPY-re-nown |
| Nama dagang | Slynd |
| Nama lain | Dihidrospirenon; Dihidrospirorenon; 1,2-Dihidrospirorenon; MSp; SH-470; ZK-30595; LF-111; asam 17β-Hidroksi-6β,7β:15β,16β-dimetilena-3-okso-17α-pregn-4-ena-21-karboksilat, γ-lakton |
| AHFS/Drugs.com | |
| License data |
|
| Rute pemberian | Oral[1] |
| Kelas obat | Progestogen; Progestin; Antimineralocorticoid; Steroidal antiandrogen |
| Kode ATC | |
| Status hukum | |
| Status hukum | |
| Data farmakokinetika | |
| Bioavailabilitas | 66–85%[1][5][6] |
| Pengikatan protein | 95–97% (menjadi albumin)[4][1][5] |
| Metabolisme | Hati (kebanyakan independen dari CYP450 (reduksi, sulfasi, dan pembelahan cincin lakton), beberapa kontribusi CYP3A4)[5][7][8][9] |
| Metabolit | • Drospirenone acid[4] • 4,5-Dihydrodrospirenone 3-sulfate[4] |
| Waktu paruh eliminasi | 25–33 jam[4][5][1] |
| Ekskresi | Urine, feses[4] |
| Pengenal | |
| |
| Nomor CAS | |
| PubChem CID | |
| DrugBank |
|
| ChemSpider |
|
| UNII | |
| KEGG |
|
| ChEBI | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.060.599 |
| Data sifat kimia dan fisik | |
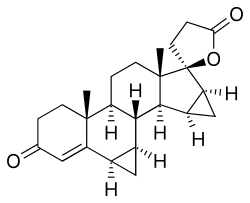
| Rumus | C24H30O3 |
| Massa molar | 366,50 g·mol−1 |
| Model 3D (JSmol) | |
| |
| |
| (verify) | |
Drospirenon adalah obat progestin dan antiandrogen yang digunakan dalam pil kontrasepsi oral gabungan untuk mencegah kehamilan dan terapi hormon menopause.[1][10] Obat ini tersedia baik sebagai sediaan tunggal maupun dalam kombinasi dengan etinilestradiol.[10][4] Obat ini merupakan analog dari spironolakton.[11] Obat ini diminum.[1][4]
Efek samping yang umum termasuk jerawat, sakit kepala, nyeri payudara, peningkatan berat badan, dan perubahan menstruasi. Efek samping yang jarang terjadi mungkin termasuk kadar kalium tinggi dan pembekuan darah (bila dikonsumsi sebagai pil kombinasi estrogen-progestogen), antara lain.[4][12] Drospirenon adalah progestin atau progestogen sintetis, dan karenanya merupakan agonis reseptor progesteron, yakni target biologis progestogen seperti progesteron. Obat ini memiliki aktivitas antimineralokortikoid dan antiandrogenik tambahan dan tidak memiliki aktivitas hormonal penting lainnya.[1] Karena aktivitas antimineralokortikoidnya dan kurangnya aktivitas off-target yang tidak diinginkan, drospirenon dikatakan lebih mirip dengan progesteron bioidentik daripada progestin lainnya.[13][14]
Drospirenon dipatenkan pada tahun 1976 dan diperkenalkan untuk penggunaan medis pada tahun 2000.[15][16] Obat ini tersedia secara luas di seluruh dunia.[10] Obat ini terkadang disebut sebagai "progestin generasi keempat".[17][18] Obat ini tersedia sebagai obat generik.[19]